 
Each isotope has an abundance of 78.70%, 10.13%, and 11.17%, respectively. Magnesium has three naturally occurring isotopes: 24Mg, 25Mg, and 26Mg. We can start by using magnesium as an example. Using isotope abundance to calculate atomic weightĪs stated previously, the number of isotopes and their percent abundance are all that are needed to calculate the atomic weight of an element. If given the atomic mass of the isotopes of an element as well as their relative abundances, we can follow simple steps to calculate the atomic weight. There are many isotopes that occur much more commonly than others, and therefore have a greater impact on the atomic weight. Though there may be many naturally occurring isotopes of an element, they do not exist in equal amounts. This is where isotope abundance comes in. Atomic weight on the other hand is the weighted average of all of the isotopes of an element that exist. This is solely the calculation of the weight of protons and neutrons in amu. Atomic mass is defined as the mass of an individual atom of an element.  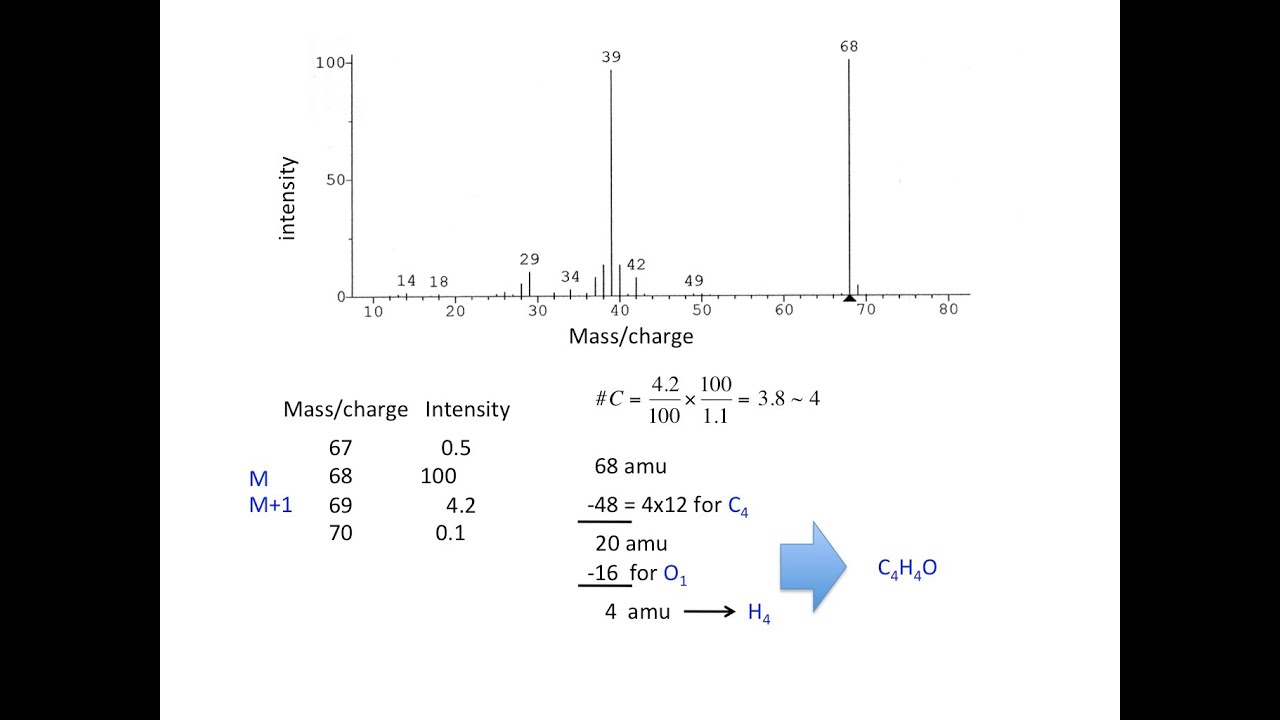
Though they sound like synonyms, atomic mass and atomic weight are different. As both protons and neutrons make up an atom’s mass, when an element differs in its number of neutrons, it is impactful on the mass. Electrons are an important part of elements as well, but they have such a small mass that they are considered negligible when calculating atomic mass. How does isotope abundance impact atomic weight?Ītomic mass depends on the composition of protons and neutrons in an element, with each weighing 1 atomic mass unit (amu). Carbon-14 is a naturally occurring carbon isotope that radioactively decays. One isotope of carbon, carbon-14, defies the normal reactivity of the stable element. Carbon is known to be a very stable element, often being involved in predictable reactions. In some instances, isotopes can have different reactivity, but in most cases, the defining difference is the number of neutrons.Ī common example of an isotope having reactivity that differs from what the element is known for is carbon. Though these two versions of the same element differ in the number of neutrons, it is important to note that they do not differ in the number of protons and electrons. Isotopes are very similar versions of the same element, only having one difference: the number of neutrons. Neutron: Neutrally charged subatomic particle located in the nucleus of an atom.Proton: Positively charged subatomic particle located in the nucleus of an atom.Isotope: when an element has a different form in which it contains the same number of protons, but differs in the number of neutrons.Quantifying Protons, Neutrons, and Electrons.In this tutorial, we will learn what isotope abundance is and how to use it to calculate the atomic weight of an element. If you look closely, it is clear that these values are almost never whole numbers. Molecular M ass (M r ) is the sum of all the relative atomic masses for all the atoms in a given formula.įor covalent compounds it is called the Relative Molecular Mass.įor ionic compounds it is called the R elative F ormula Mass.When looking at the periodic table, each element has a value displayed for the atomic mass. There is no unit as it is a relative value. The relative atomic mass of Copper is therefore ( 70/ 100 x 63) + ( 30/ 100 x 65) = 63.6 The proportions of each isotope are 69.17% and 30.83%. The proportion of each isotope in the element.Ĭopper has 2 isotopes, 63Cu and 65Cu. The relative isotopic mass of each isotope. To find the relative atomic mass of an element we must know: However, most elements contain a mixture of isotopes, each with a different mass number. Relative isotopic mass is then simply the mass number of the isotope, e.g. I n a pure isotope all the atoms have the same atomic structure and the same mass. take both the mass of a proton and a neutron as being equal to 1 u.neglect the tiny contribution to atomic mass from electrons.Therefore, 1 u is 1/ 12th the mass of an atom of carbon-12. By international agreement the mass of an atom of carbon-12 is given as 12 unified atomic mass units (u). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
